Abstract
Background: The efficacy of spike messenger RNA vaccines against SARS-CoV-2 in immunocompetent individuals is about 95%, while in immunocompromised patients (pts) no unique value has been established, probably due to a dysregulation of the immune system. Most patients with chronic lymphocytic leukaemia (CLL) present a consistent immunological dysfunction, which increases the risk of mortality and morbidity from infections, including SARS-CoV-2. Moreover, CLL pts have shown a highly heterogeneous response to vaccinations, including the one for SARS-CoV-2. It depends on many factors, such as clinical features, disease and treatment status, types of therapy, vaccine administered, and immunoglobulin level at time of vaccination.
Aims: We analysed the antibody responses rate and the efficacy of SARS-CoV-2 2 vaccine in our unselected population, stratifying pts according to treatment status (treated vs naive) and type of treatment (chemo-immunotherapy-CIT, BTK or BCL-2 inhibitors, eventually associated with monoclonal antibodies (MoAb).
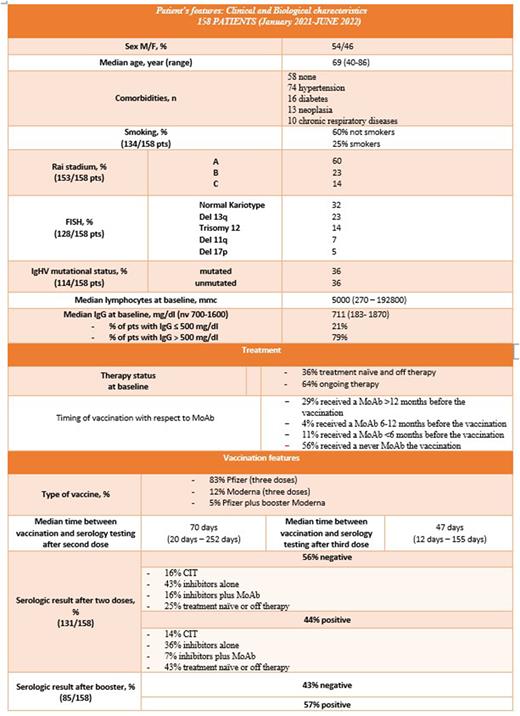
Patients and methods: This retrospective study included 158 CLL pt s from Gemelli Hospital, Rome and Businco Hospital, Cagliari, who received two or three doses of mRNA vaccine (BNT162b2 or mRNA-1273) between January 2021 and June 2022. Baseline demographics, treatment history and laboratory parameters including lymphocyte count and IgG levels prior to first dose of COVID-19 vaccine were collected and pts were tested for SARS-CoV-2 anti-spike Ig at different time points. Median age at time of vaccination was 69 yrs (range 40-86); 54% pts were males; 62% had significant comorbidities (hypertension, diabetes, neoplasia,COPD). 57% of pts received at least one line of therapy, CIT or BTKi or BCL-2i, with a median number of 1 (range 0-6) and 64% pts were on active treatment. CLL treatment was not held or modified at the time of vaccination. Median IgG level at baseline was 711 mg/dL, with IgG > 500 mg/dL in 79% of pts. BNT162b2 was administered to 83%, mRNA-1273 to 12%, two BNT162b2 and mRNA-1273 booster to 5%. (Table 1).
Results: At the time of first serologic testing (median 70 days after the 2nd dose), 56% pts tested negative and 44% positive. Among pts who did not develop serological response, 43% were on treatment with inhibitors alone, 25% were off therapy or treatment-naive, 16% were on CIT and 16% were treated with inhibitors plus MoAb. Among patients who developed humoral response 36% were on BTKi or BCL2i, 14% were on CIT, 7% were on treatment with inhibitors plus MoAb, 43% were not treated at the time of vaccination. 89% of pts that received MoAb less than 6 months before vaccination failed to develop humoral response, with 67% of non-responders if treated in the previous 6-12 months. At the second serologic testing, (median of 47 days after the booster), 57% patients tested positive, but we unfortunately missed data from 43% of population, so we hypothesize an underestimation of the real humoral response rate. Furthermore, 30% of patients negative after the first and second vaccine doses developed a humoral response after the booster. On the other hand, 1 patient responder after first and second dose lost humoral response after booster, due to concomitant MoAb administration (Tab.1). We reported only 12 SARS-CoV-2 infections after vaccination, with 2 deaths due to COVID-19 in non-responders pts. No patient developed severe adverse reactions to the vaccine.
Conclusions: Our experience confirms a low humoral response in CLL, due to both disease and therapy related factors. Patients either naïve or off-therapy at the time of vaccination yielded better results, probably due to a lower disease burden. We confirm that timing of MoAb administration strongly influences the response rate. Additionally, we know that BTKi impair the humoral response to any vaccination and so also to the one against SARS-CoV-2. Our data show that the third dose confers higher rates of response and a wider protective effect in CLL pts. In conclusion, vaccination is strongly recommended to all CLL pts. Due to the higher infectious risk caused by CLL immune dysregulation, it is important to strictly evaluate the timing of vaccination and maintain safety measures at any time of both disease and pandemic waves.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal